  
lines, when these excited electrons relax to their ground state energy levels. , N# where #N# is an integer), they give rise to discrete peaks in their spectra, i.e. they correspond to the principal quantum number #n = 1, 2. It resists UV rays and will float in water. We can learn about winds in stars from this. Wet-Environment Easy-Splice Rope For Lifting Strong even when wet, this rope is a high-performance polyethylene also known as Spectra. 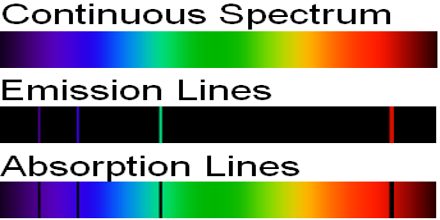
The width of the line can tell us how fast the material is moving. Since their energy levels are quantized (i.e. The spectral line also can tell us about any magnetic field of the star. Some of their highest-energy electrons can get excited by photons of specific energies to an excited state. Altogether, Spectraline products are used in a wide variety of applications such as monitoring molten glass, hot steel and aluminum ingots, fault detection in high speed plastic webs and moving parts, characterizing automotive and aerospace engine combustion, pool fires and rocket engine plumes. IR peaks have width to them because the dipole moment changes while the molecule stretches and bends, changing their electron distribution and altering their receptiveness to the incoming light source the integral under their peaks is nonzero and not small.Ītomic emission spectra, on the other hand (at least the kind that you're referring to), receiving a coherent/focused light source to excite the atom, generally have little to no width to their lines. The stretches and bends of bonds alter the dipole moment of the molecule, which is directly proportional to the strength of the IR peak. Its peaks arise from the vibrational modes of molecules, not atoms. We record high resolution spectra of different HCLs using a Fourier transform spectrograph: (i) UNe, UAr, ThNe, and ThAr lamps in the spectral range from. Because atoms have quantized energy levels. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
